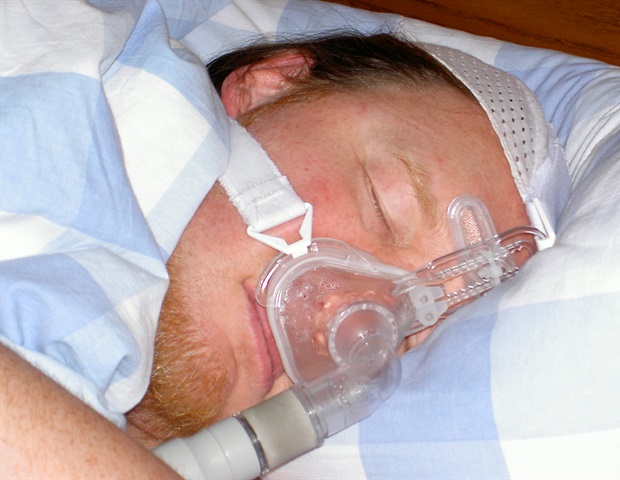
New research showed that tirzepatide, the compound in Zepbound, improved symptoms of obstructive sleep apnea.
Vous n'êtes pas connecté
- English
- Français
- عربي
- Español
- Deutsch
- Português
- русский язык
- Català
- Italiano
- Nederlands, Vlaams
- Norsk
- فارسی
- বাংলা
- اردو
- Azərbaycan dili
- Bahasa Indonesia
- Հայերեն
- Ελληνικά
- Bosanski jezik
- українська мова
- Íslenska
- Türkmen, Түркмен
- Türkçe
- Shqip
- Eesti keel
- magyar
- Қазақ тілі
- Kalaallisut ; kalaallit oqaasii
- Lietuvių kalba
- Latviešu valoda
- македонски јазик
- Монгол
- Bahasa Melayu ; بهاس ملايو
- ဗမာစာ
- Slovenščina
- тоҷикӣ ; toğikī ; تاجیکی
- ไทย
- O'zbek ; Ўзбек ; أۇزبېك
- Tiếng Việt
- ភាសាខ្មែរ
- རྫོང་ཁ
- Soomaaliga ; af Soomaali
 Maroc - ASIANEWSDAY.COM - A la Une - 21/Jun 20:29
Maroc - ASIANEWSDAY.COM - A la Une - 21/Jun 20:29
Eli Lilly expects FDA decision on weight loss drug Zepbound for sleep apnea as early as end of the year
Eli Lilly on Friday said it applied for U.S. approval of its weight loss drug Zepbound for the treatment of the most common sleep-related breathing disorder and expects regulators to make a decision as early as the end of the year. If cleared by the Food and Drug Administration, the company plans to launch Zepbound for so-called obstructive […]
Articles similaires
Is Eli Lilly (LLY) a Buy on Tirzepatide Success in Sleep Apnea?
Eli Lilly (LLY) files for the approval of tirzepatide to treat obstructive sleep apnea in adults with obesity in the United States. If approved, sales...
Tirzepatide emerges as first drug therapy for obstructive sleep apnea
Researchers at University of California San Diego School of Medicine and international collaborators have led a worldwide, advanced study...
Scientists find first drug to treat sleep apnea
In recent research led by the University of California San Diego School of Medicine, scientists have found a new potential treatment for obstructive...
Eli Lilly (LLY) Gets FDA Nod for Alzheimer's Drug Donanemab
Eli Lilly's (LLY) Alzheimer's disease drug, Kisunla (donanemab), wins FDA approval. Kisunla will be the second drug on the market to treat the...
SEC recommends grant of permission to import and market Eli Lilly's weight loss drug
The Subject Expert Committee (SEC), which advises the national drug regulator on drugs and trials approval matters, has recommended grant of...
FDA Approves Donanemab, Eli Lilly’s New Drug for Early Alzheimer’s
The drug, Kisunla, made by Eli Lilly, is the latest in a new class of treatments that could modestly slow cognitive decline in initial stages of the...
Weight loss drug could ease sleep apnoea in people with obesity, study says
Research raises possibility that tirzepatide could become first pharmaceutical treatment for the sleeping disorder
FDA Approves Kisunla (donanemab-azbt) for the Treatment of Early Symptomatic Alzheimer's Disease
INDIANAPOLIS, July 2, 2024 /PRNewswire/ -- The U.S. Food and Drug Administration (FDA) approved Kisunla™ (donanemab-azbt, 350 mg/20 mL...
Lifeline for Obese Sleep Apnea Patients
Cleveland Clinic study reveals that weight loss surgery significantly reduces the risk of heart complications by 42% and the risk of death by 37% in...
Les derniers communiqués
-
Seatrade Maritime Awards Extends Entry Deadline to 8th September 2023
Informa markets - 28/08/2023